Effectors, predominantly of proteinaceous in nature are secretions by pathogenic bacteria and, filamentous oomycete and fungi intended to neutralise the first layer of defense termed pattern-triggered immunity (PTI) also termed as basal immunity in the two-layered plant defense strategy. While in the second layer of defense, effectors are recognised by the host resistance proteins directly or indirectly for activating the plant responses against the invader called the effector-triggered immunity (ETI). Strikingly, the functional aspects of only a smaller number of effectors are known providing opportunities to further pursue studies on manipulating the defense pathways for desired advantages in crop production. Park et al. (2012) has set the stage by elegantly implicating ubiquitin proteosome system (UPS) in a two-way reciprocal regulation of a pathogen effector and its target protein leading to suppression or lessen the interference on host PTI in rice-blast fungus (Magnaporthe oryzae) pathosystem.
In natural environments, plants will continuously experience a low biotic stress due to exposure to numerous microbial pathogens, and they incur costs for using the constitutional defences (basal immunity) to ward off the pathogens. It becomes essential to balance between immunity and plant growth even in normal conditions. Gao et al. (2021) lead by Zuhua He, have exquisitely brought to light a conserved immune suppression network in rice-blast pathosystem, and appropriately fits this into the genetic and biochemical context of blast resistance (Figure 1). The pathogen-secreted effector, AvrPiz-t and its mimic, a host susceptibility protein, RESISTANCE OF RICE TO DISEASES 1 (ROD1) homeostatically regulate the levels of reactive oxygen species (ROS) including hydrogen peroxide in the cellular environment arising in response to pathogenesis. This has been dealt in an earlier DeepView blog in this site, “Balancing trade-off between disease resistance and plant growth”. This controlled nature of resistance expression is mediated by ubiquitin-dependent pathways of protein degradation (ubiquitin proteasome system, UPS). UPS is an essential post-translational regulatory mechanism important for protein turn over in cells and is needed for several homeostatic functions. Functioning of the UPS is carried out by three enzymes, ubiquitin-activating enzyme, (E1), ubiquitin-conjugating enzyme (E2), and ubiquitin-protein ligase (E3).
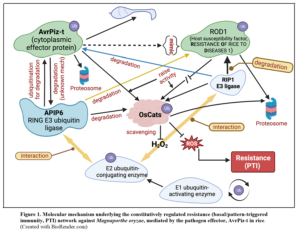 Catalase (CAT) helps in scavenging reactive oxygen species including H2O2 during stress. The plant susceptibility factor, ROD1 recruits and hikes CatB activity to eliminate H2O2 resulting in limited immune responses in favour of plant growth. Blocking the ROD1 functions leads to activation of the physiologically expensive constitutive immune responses conferring broad-spectrum resistance to multiple diseases (including sheath blight and bacterial blight, besides blast) at the expense of growth. Further, experimentation with transgenic lines, chromosome segment substitution lines, mRNA and protein levels assessment, and tobacco cell death analysis indicates that rod1 (natural variant of ROD1 discovered by Gao et al. 2021) modifies the effect of catalase activation facilitating immune regulation. By regulating ROS-scavenging causing immunity suppression, ROD1 equilibrates immunity and plant growth fitness under normal conditions. These results further are suggestive of the principle that ROD1, ROS metabolism and protein homeostasis regulated by UPS might be of a significant hub for balancing immunity and plant growth. Examination of this allele frequency in different rice varieties showed its adaptation to indica rice species of tropical and subtropical (low latitude) regions. Transgenic seeds carrying rod1 allele are available with Gao et al. (2021, corresponding author: Zuhua He, zhhe@cemps.ac.cn) for sharing upon request with Materials Transfer Agreement.
Catalase (CAT) helps in scavenging reactive oxygen species including H2O2 during stress. The plant susceptibility factor, ROD1 recruits and hikes CatB activity to eliminate H2O2 resulting in limited immune responses in favour of plant growth. Blocking the ROD1 functions leads to activation of the physiologically expensive constitutive immune responses conferring broad-spectrum resistance to multiple diseases (including sheath blight and bacterial blight, besides blast) at the expense of growth. Further, experimentation with transgenic lines, chromosome segment substitution lines, mRNA and protein levels assessment, and tobacco cell death analysis indicates that rod1 (natural variant of ROD1 discovered by Gao et al. 2021) modifies the effect of catalase activation facilitating immune regulation. By regulating ROS-scavenging causing immunity suppression, ROD1 equilibrates immunity and plant growth fitness under normal conditions. These results further are suggestive of the principle that ROD1, ROS metabolism and protein homeostasis regulated by UPS might be of a significant hub for balancing immunity and plant growth. Examination of this allele frequency in different rice varieties showed its adaptation to indica rice species of tropical and subtropical (low latitude) regions. Transgenic seeds carrying rod1 allele are available with Gao et al. (2021, corresponding author: Zuhua He, zhhe@cemps.ac.cn) for sharing upon request with Materials Transfer Agreement.
Maintaining how AvrPiz-t targeting directly APIP6 (AvrPiz-t INTERACTING PROTEIN 6) regulates ROS levels to modulate rice immunity is not clear, in another very recent publication, You et al. (2022) led by Yuese Ning further demonstrate the direct interaction between APIP6 and OsCATC both in vitro and in vivo (rice) and pinpoints the fact that AvrPiz-t also stimulates OsCATC-mediated H2O2 scavenging without intervening factors. For counteracting this, E3 ubiquitin ligase, APIP6 targets both AvrPiz-t and OsCATC for degradation for increasing ROS levels for maintaining active defense against the pathogen. This leads to a suggestion that APIP6-promoted degradation of the experimented OsCATC in vivo is devoid of any feedback regulation on APIP6. This underpins the importance of catalase in negative regulation of rice immunity. These authors show that the mutants oscatc and CR-catb (CRISPR genome-edited at CATB), despite with increased resistance contributed by higher ROS levels is not devoid of growth penalties. Rice genome encodes three forms of catalase (CAT) genes, OsCATA, OsCATB, and OsCATC, and all are targeted for degradation in the tightly controlled immunity network. These three forms of CAT genes may function either as a homologous or heterologous complex. Next step could focus on dissecting out the involvement of the CAT gene complexity for exploiting them for developing plants with enhanced resistance without compromising yield attributes. Further, the trait of accommodating the pathogen for a certain level of the disease without causing any negative effect on growth and grain yield is preferred for preventing or slowing down the gain of virulence by the pathogen in the arms race between plant and pathogen, as selection pressure on the pathogen is expected to be nil to minimum, thus enhancing the stability of the defense strategy (Pagan and Farcia-Arenal 2020). For this reason, the rod1 genotype would be a suitable model for studies on genetic regulation and molecular basis of negative effects of disease, characteristic of tolerance to diseases.
References
Gao, M., He, Y., Yin, X., Zhong, X., Yan, B., Wu, Y., Chen, J., Li, X., Zhai, K., Huang, Y., Gong, X.,
Chang, H., Xie, S., Liu, J., Yue, J., Xu, J., Zhang, G., Deng, Y., Wang, E., Tharreau, D., Wang,
F-L., Yang, W., He, Z. (2021) Ca2+ sensor-mediated ROS scavenging suppresses rice
immunity and is exploited by a fungal effector. Cell 184, 5391-5404.
Pagan, I., Farcia-Arenal, F. (2020) Tolerance of Plants to Pathogens: A unifying view. Annu. Rev.
Phytopathol. 58, 77-96.
Park, C-H., Chen, S., Shirsekar, G., Zhou, B., Khang, CH., Songkumarn, P., Afzal, AJ., Ning, Y.,
Wang, R., Bellizzi, M., Valent, B., Wang, G-L. (2012) The Magnaporthe oryzae effector
AvrPiz-t targets the RING E3 ubiquitin ligase APIP6 to suppress pathogen-associated molecular
pattern-triggered immunity in rice. Plant Cell 24, 4748-4762.
You, X., Zhang, F., Liu, Z., Wang, M., Xu, X., He, F., Wang, D., Wang, R., Wang, Y., Wang, G,
Chu, C., Wang, G-L. Ning, Y. (2022) Rice catalase OsCATC is degraded by E3 ligase
APIP6 to negatively regulate immunity. Plant Physiol. 190, 1095-1099.
Acknowledgement: Critical examination of the graphical summary by Dr. Ruyi Wang, State Key Laboratory for Biology of Plant Diseases and Insect Pests, Institute of Plant Protection, Chinese Academy of Agricultural Sciences, Beijing, China is gratefully appreciated.
Suggested citation: Sridhar, R. (2022, December 07) Catalase genes, a handle for manipulating rice blast resistance. [Blog] DeepView, https://sridharr.com/catalase-genes-as-a- handle-for-manipulating-rice-blast-resistance/.


